Atypical cutaneous and mucosal lichen planus in a 53-year-old Filipino male: case report
SPMC J Health Care Serv. 2017;3(1):1 ARK: http://n2t.net/ark:/76951/jhcs69mz7x
Joanne Kate M Martinez,1 Bryan Edgar K Guevara,1 Lalaine R Visitacion,1
1Department of Dermatology, Southern Philippines Medical Center, Bajada, Davao City, Philippines
Correspondence Joanne Kate M Martinez, katemartinez620@yahoo.com
Received 03 January 2017
Accepted 10 March 2017
Cite as Martinez JKM, Guevara BEK, Visitacion LR. Atypical cutaneous and mucosal lichen planus in a 53-year-old Filipino male: case report. SPMC J Health Care Serv. 2017;3(1):1. http://n2t.net/ark:/76951/jhcs69mz7x
Abstract
Lichen planus (LP) is a rare papulosquamous inflammatory disease. We report a 53-year-old male with a 6-month history of multiple, pruritic, brown, atrophic macules on the face. He also had erythematous, pruritic, scaly papules on the trunk, lower back, genitalia and the extensor surface of the upper extremities, as well as plaques with erosions on the lower lip and buccal mucosa. The initial diagnosis of atypical LP was made through dermatoscopy and confirmed through skin punch biopsy and direct immunofluorescence. The patient was successfully treated with topical corticosteroids and chlorhexidine gluconate mouthwash initially, followed by an 8-week course of oral corticosteroids.
Keywords. Wickham striae, Max Joseph cleft, papulosquamous disease, direct immunofluorescence, dermatoscopy
Introduction
Lichen planus (LP) is a unique papulosquamous dermatosis affecting the skin, mucous membranes, nails and hair.
1 The typical lesions are faintly erythematous to violaceous, flat-topped, polygonal papules that are symmetrically and bilaterally distributed on the flexural areas of the extremities.
2 The prevalence of LP in the general population has been estimated to range from 0.1 to 4%.
3 4 Based on the Philippine Dermatological Society Health Information System, among 422,998 dermatologic consults from 2011 to 2016, 303 were cases of LP (7 per 10,000 dermatologic consults, or 0.07%).
5 The usual onset of LP lesions in two thirds of patients is within 30-60 years of age.
3 Nails may exhibit onycholysis, onychorrhexis, subungual hyperkeratosis and/or anonychia, but the classic nail finding is pterygium or the forward formation of the eponychium.
2 Dermatoscopy of a lesion usually reveals Wickham striae, which are networks of linear or pinpoint whitish structures and are pathognomonic of LP.
6 In this article, we describe the case of a 53-year old male presenting with atrophic macules on the face and scaly papules on the extensors. Dermatoscopic and histopathologic findings were consistent with LP.
Clinical features
A 53-year-old male came to our clinic with a 6-month history of gradual development of multiple, reddish, elevated, and itchy lesions on the face and extensor areas of the upper and lower extremities. He also noted gradual thickening and increased brittleness of his nails. The pruritus intensified over time, while the initially elevated lesions on the face gradually flattened. Similar reddish lesions, which eventually progressed to painful erosions, developed on the patient’s lower lip, buccal mucosa, and genitalia starting two months prior to consultation. A dermatologist prescribed clobetasol propionate 0.05% lotion, which the patient applied on the lesions twice daily. The lesions did not change, and symptoms were not resolved after one week of treatment, so the patient decided to consult us. A review of systems and the patient’s past medical history were unremarkable. The patient did not recall experiencing flu-like symptoms, sore throat or weight loss prior to the onset of the lesions. None of his immediate family members had skin cancer, other malignancies or similar skin lesions.
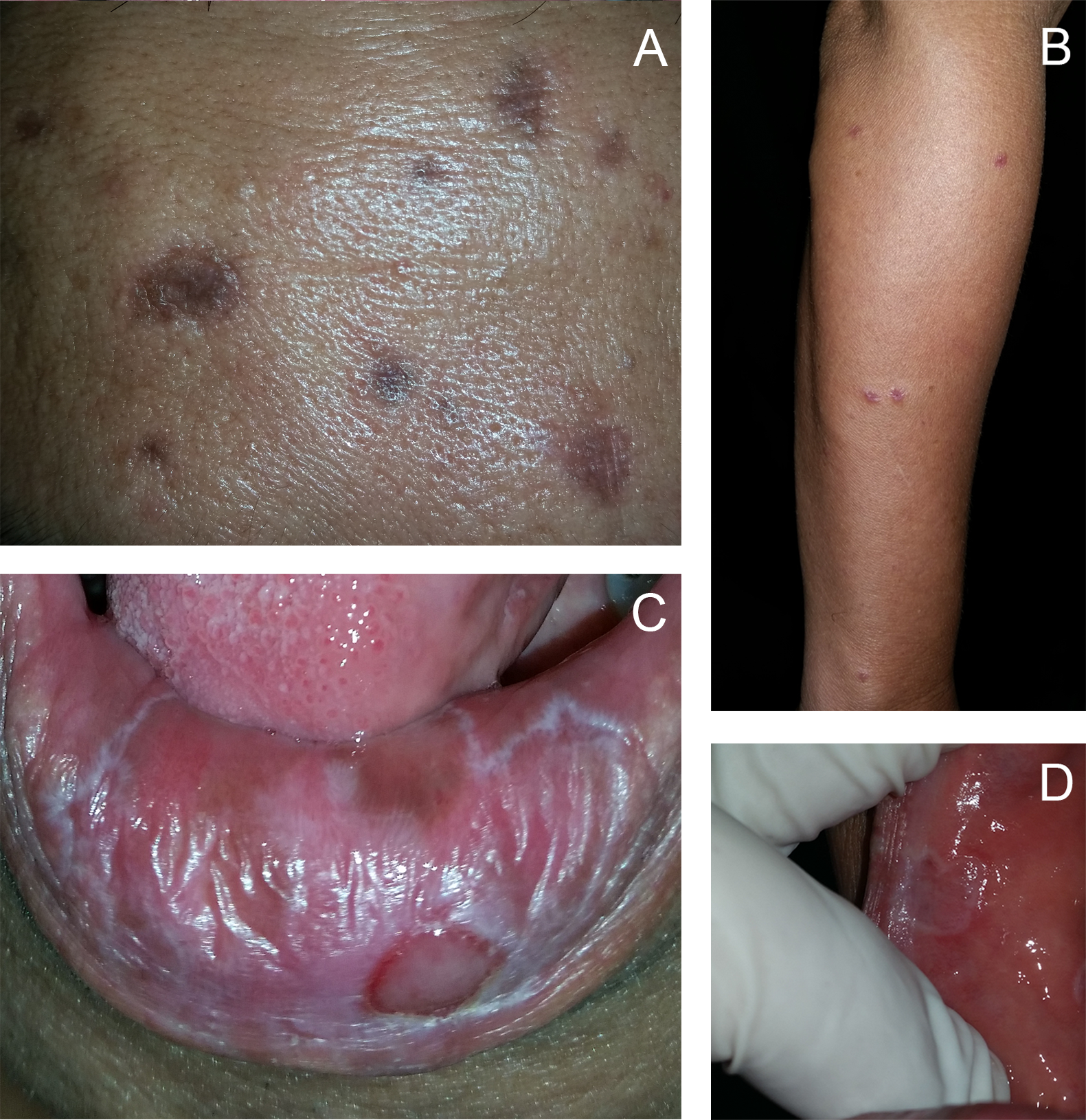
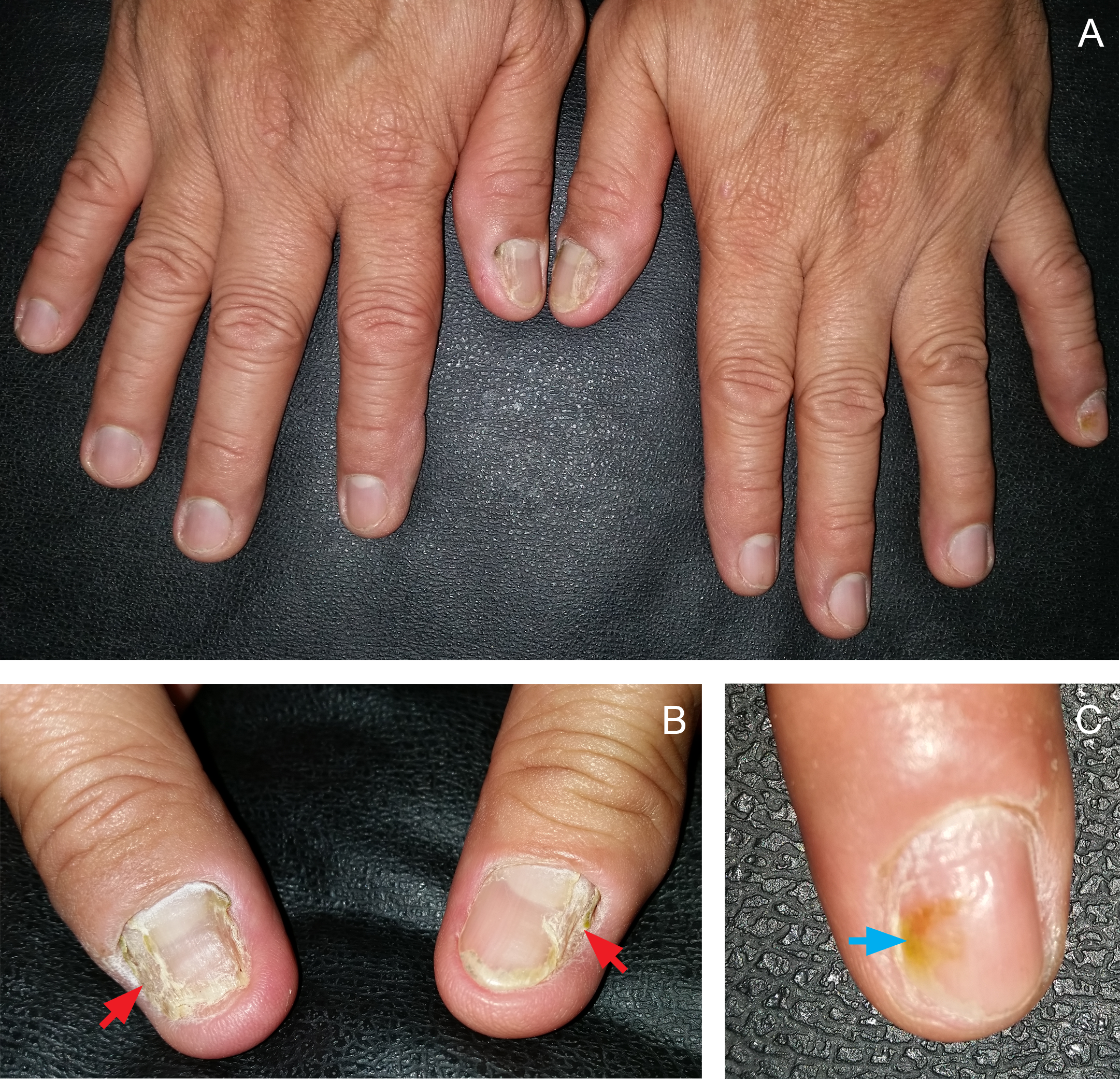
Physical examination, which centered on the skin, revealed multiple, dark brown, and discrete atrophic macules with well-defined edges on the face, as well as sparse, erythematous, well-demarcated, and symmetrically distributed papules on the trunk, lower back, and extensors of the upper extremities (Figure 1). There were also multiple, ill-defined, whitish, and reticulated plaques with erosions on the patient’s lower lip and buccal mucosa. (Figure 1). The coronal sulcus at the dorsum of the patient’s penis had erythematous papules in agminate pattern, with shiny, whitish, and thin scales over the papules. The fingernails exhibited onycholysis, onychodystrophy, chromonychia, and subungual hyperkeratosis (Figure 2). There were no apparent changes in the toenails.
|
|
Figure 1
Dark brown atrophic macules on the forehead (A), sparse erythematous papules on the extensor area of the right forearm (B), whitish reticulated plaque with erosion on the lower lip (C), and eroded plaque with well-defined whitish border on the buccal mucosa (D).
|
|
|
Figure 2
Involvement of the fingernails of both hands (A), with onychodystrophy (B: red arrows), and chromonychia (C: blue arrow).
|
Diagnostic approaches
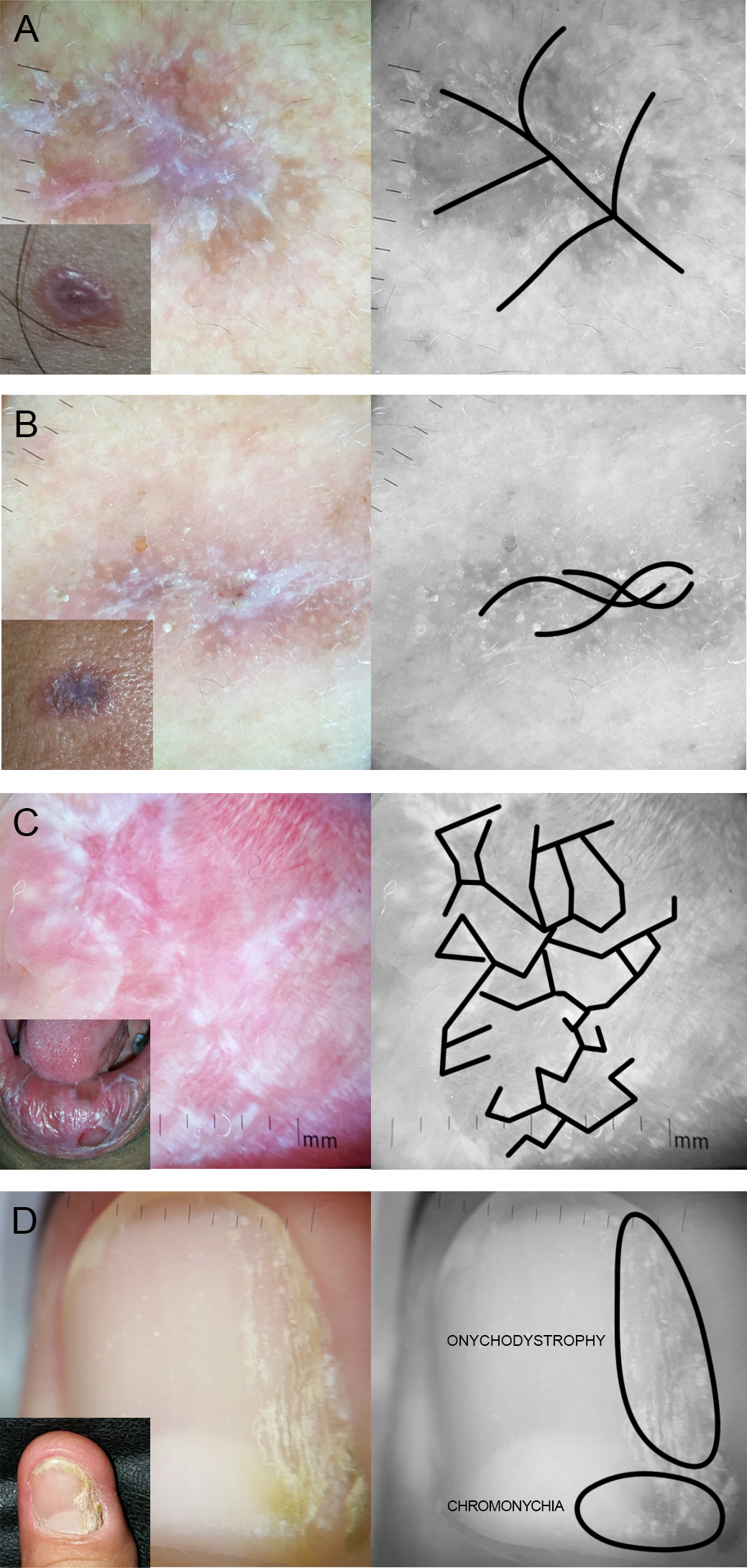
We examined several lesions under a dermatoscope. Lesions from the forehead, lips, and legs had Wickham striae—polymorphic pearly white lines arranged in tree-like, lace-like or fern leaf pattern (Figure 3). The fingernails exhibited subungual hyperkeratosis, onycholysis, destruction of the nail plate, and chromonychia. Because the presence of Wickham striae on papules, macules, and plaques is characteristic of LP,
2 our subsequent diagnostic approach was geared towards confirming the diagnosis.
|
|
Figure 3
Dermatoscopic (colored large photos; x10) and gross (inset photos) views of the patient’s lesions: an erythematous papule from the leg showing fine white lines (Wickham striae) in fern leaf pattern (A); an atrophic macule on the forehead showing linear Wickham striae (B); a whitish reticulated plaque on the lower lip with arboriform Wickham striae (C); and a fingernail with longitudinal onychodystrophy and chromonychia (D).
|
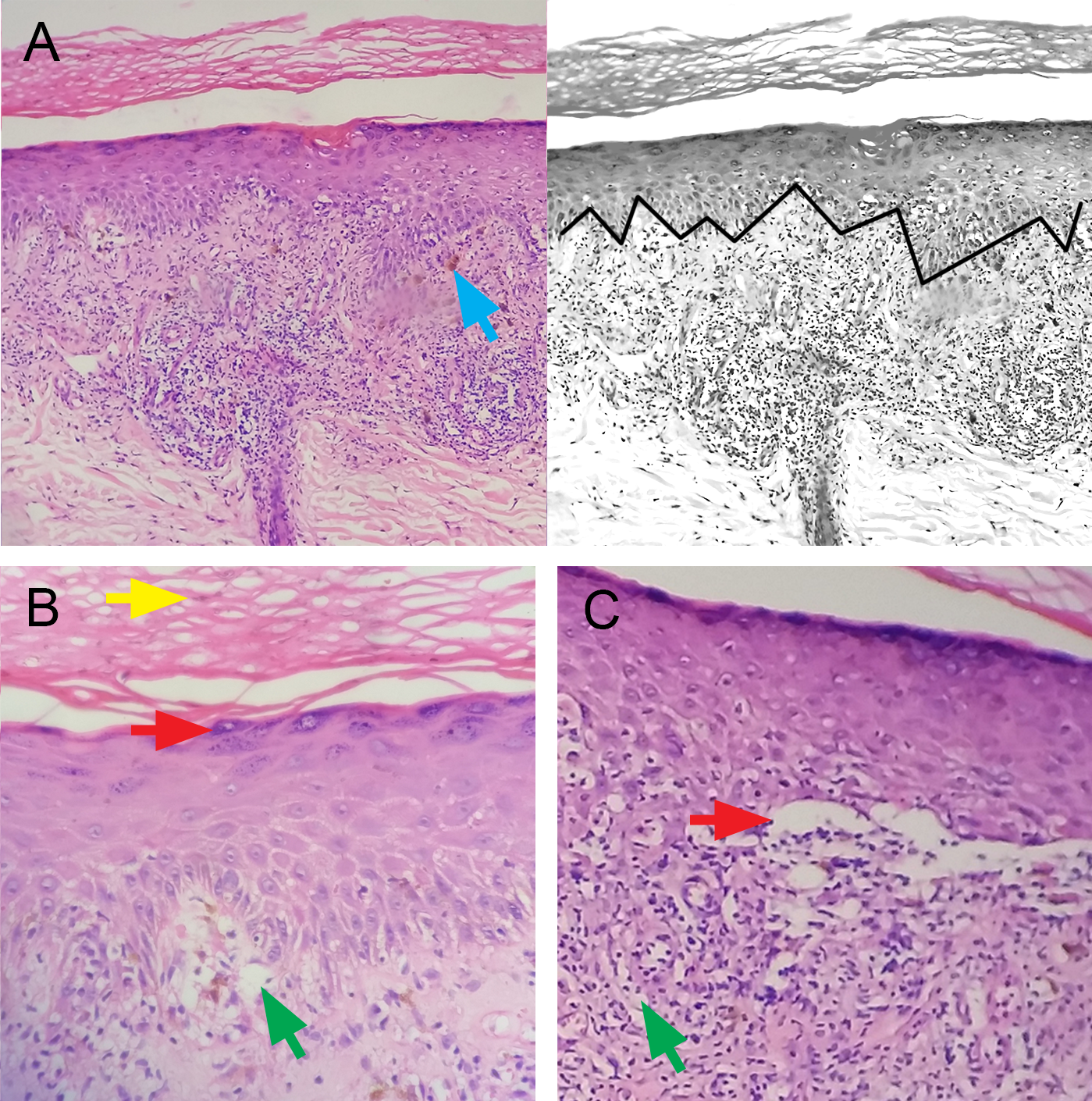
Skin punch biopsies from an erythematous papule on the left leg and from a purple atrophic patch on the left mandibular area (Figure 4) revealed basket weave orthokeratosis of the epidermis, epidermal hyperplasia (acanthosis), thickened stratum granulosum (hypergranulosis), sawtooth configuration of the rete ridge, and vacuolar degeneration of the basal layer. A horizontally elongated subepidermal space (Max Joseph cleft) and melanophages indicating pigment incontinence could be appreciated in one section of the papule sample. Lichenoid lymphohistiocytic inflammatory infiltrates were also present in the dermis. We also sent a sample of perilesional skin from an erythematous papule on the left leg for direct immunoflourescence. Results showed thick shaggy deposits of fibrinogen in the basement membrane (Figure 5). All these microscopic findings were consistent with our initial working diagnosis of LP.
|
|
Figure 4
Photomicrograph of skin biopsy showing sawtooth configuration of rete ridges (A), melanophages (A: blue arrow) in the upper dermis, indicating pigment incontinence, hyperkeratosis (B: yellow arrow), hypergranulosis (B: red arrow), vacuolar degeneration (B: green arrow), Max Joseph cleft (C: red arrow), and lichenoid lymphohistiocytic inflammatory infiltrates (C: green arrow). Hematoxylin-eosin stain, A: x10, B and C: x40.
|
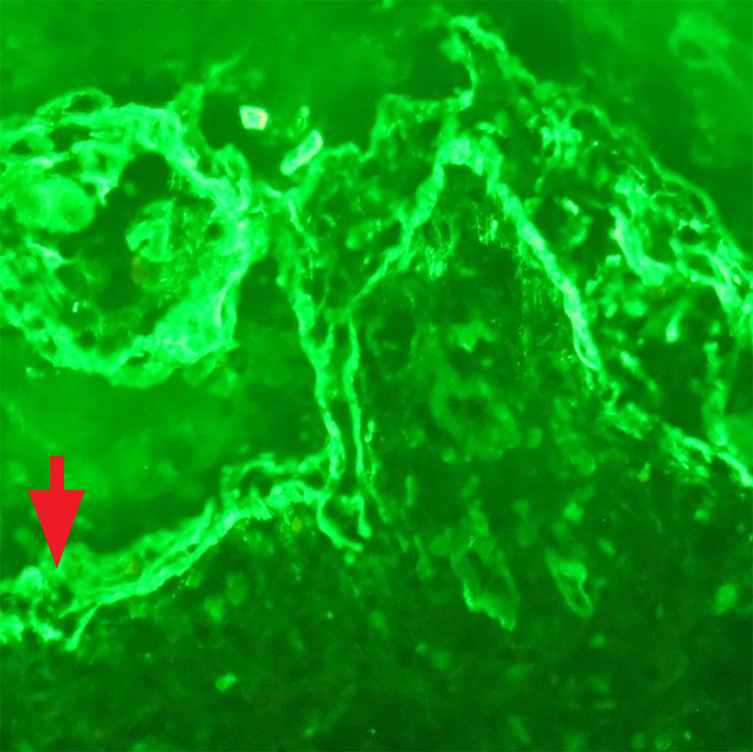
|
|
Figure 5
Direct immunofluorescence of perilesional skin (x40) showing thick shaggy deposits of fibrinogen at the basement membrane zone (red arrow).
|
Therapeutic approaches and outcomes
We instructed the patient to apply halobetasol propionate 0.05% cream twice a day for 2 weeks on the cutaneous lesions, to take oral cetirizine 10 mg daily for two weeks for the pruritus, and to gargle with chlorhexidine gluconate mouthwash thrice a day for two weeks for the oral lesions. We advised the patient to come back to our clinic after 2 weeks for reassessment, but he was not able to comply. The patient returned to our clinic after 1 month and claimed to have applied halobetasol propionate 0.05% cream and taken cetirizine as prescribed for two weeks. He also told us that, upon noting that the number of the cutaneous lesions decreased on the second week of treatment, he decided to apply halobetasol propionate 0.05% cream only intermittently. He also took cetirizine less frequently, and only when the pruritus was unbearable. On the other hand, when the patient noted a decrease in the size of the lesions on the buccal mucosa and lower lip after one week, he stopped the chlorhexidine gluconate mouthwash.
One month after initiating therapy, our initial approach only provided partial response. The lesions decreased in number but remained erythematous, and the patient reported that pruritus persisted, albeit with lesser intensity. The whitish plaques with erosions on the lower lip and buccal mucosa decreased in size. We started the patient on oral prednisone, starting at 40 mg daily (0.5 mg/kg/day), then tapered every week—with dosage decrements of 5 mg—over a total period of eight weeks.
After one week of oral prednisone therapy, the cutaneous lesions on the extremities, trunk and face further decreased in number, and pruritus was reported to have decreased to a minimum, as well. The erosions and white plaques on the lower lip and buccal mucosa disappeared completely. There were no significant changes in the nail lesions. We offered intralesional steroid injection for the fingernails, but the patient refused. After eight weeks of prednisone, the cutaneous lesions on the extremities and trunk resolved completely. Itching was completely absent, as well, but the lesions on the face left residual hyperpigmentation. We instructed the patient to return to our clinic whenever new signs and symptoms would develop.
Discussion
LP is characterized by two pathophysiologic mechanisms, namely—basal epidermal keratinocyte damage and lichenoid-interface lymphocytic reaction.
2 The exact cause of LP is unknown, although T cell-mediated pathologic alterations involving proinflammatory and counterregulatory mechanisms function in the pathogenesis of LP.
2 These T lymphocytes are directly activated by antigen binding to keratinocyte major histocompatibility complex 1 (MHC-1) or through helper lymphocytes. The production of tumor necrosis factor alpha or granzyme B subsequently induces apoptosis of basal keratinocytes.
7
The classic lesions of LP are purple, polygonal, and pruritic papules or plaques, which develop on flexural areas.
2 The lesions are usually symmetrically distributed and typically favor the wrists, ankles, shins, trunk, and sacral region, as well as the flexural surfaces of the forearms and legs, and the dorsal surfaces of the hands.
2 Lesions may also be present in other cutaneous and even mucosal sites but, interestingly, the face is usually spared in classical LP.
8 Usually, the fingernails also exhibit morphologic changes. Scalp involvement can produce scarring alopecia.
2
Our patient, however, presented with sparse erythematous papules on the extensors, atrophic macules on the face, a large plaque with erosion in the lower lip, an erosion in the buccal mucosa, erythematous agminate papules in the penis, and fingernail changes that include onycholysis and chromonychia. The dermatoscopic and histologic features of the lesions were consistent with LP. All these made us think that the patient had atypical LP.
The clinical variants of LP differ from the classic type in terms of morphology of the lesions and sites of involvement. Atypical LPs include the atrophic, hypertrophic, vesiculobullous, actinic, annular, linear, and follicular variants, as well as LP pigmentosus and LP pigmentosus inversus.
9 In the atypical atrophic variant of LP, the usual papules of classic LP are replaced by flat violaceous macules, which are usually found in the axillae and glans penis.
10 Atrophic LP is characterized by sparse distribution of lesions.
9 11 It has also been reported that LP affects the male genitalia in 25% of cases.
2 Lesions are typically present on the glans penis and would frequently show an annular pattern,
4 but in our patient, erythematous papules with thin and whitish scales and in agminate pattern could be seen in the coronal sulcus of the penis, just below the glans.
As in our patient, LP typically manifests more in fingernails than in toenails.
2 Early manifestations of affected nails include pitting and trachyonychia, while advanced disease can produce chromonychia, lamina fragmentation, onycholysis and splinter hemorrhages.
8 Affected nails may also present with longitudinal ridges, onychorrhexis, and distal splitting. Pterygium, a pathognomonic LP nail finding, was not observed in our patient.
8
The rarity of some LP variants and their atypical presentations make their timely diagnosis and management more difficult in the clinical setting. Viewing the lesions through an office dermatoscope often establishes the diagnosis of LP. The diagnostic accuracy of dermatoscopy is superior to clinical examination alone in the field of inflammatory dermatoses, since dermatoscopy sharply demonstrates blood vessel morphology and distribution, background color, surface scales and follicular disturbances within or around a lesion.
12 The presence of Wickham striae—or the whitish structures with reticulated, arboriform or fern leaf pattern over papules, plaques or macules—is pathognomonic of LP.6 In lesions on the lip or buccal mucosa, Wickham striae appear tree-like or in the form of a lacy network.
6 At the borders of the white streaks of Wickham striae, linear vessels (radial capillaries) and erythematous globules may be observed.
6 In our patient’s case, the diagnosis of LP was clinched through our findings of several patterns of Wickham striae over the lesions upon dermatoscopy.
Histopathology confirms the diagnosis of LP, because findings in all forms of LP remain consistent across the many variants of the condition.
8 Florid hyperkeratosis, wedge-shaped hypergranulosis, and irregular sawtooth acanthosis of the rete ridges are typically seen in light microscopy of LP lesions. A histologic finding of compact orthokeratosis above zones of wedge-shaped hypergranulosis manifests as the whitish linear streaks in Wickham striae that can be appreciated under the dermatoscope.
6 The dermoepidermal junction usually exhibits vacuolar degeneration, while the superficial dermis can have band-like lymphocytic infiltrates. Apoptotic keratinocytes or civatte bodies are seen in both the epithelium and upper dermis.
8 Occasionally, Max Joseph spaces, which are subepidermal clefts formed by acantholysis or hydropic degeneration of basal cells, can be appreciated under light microscopy.
13 Immunohistochemistry usually reveals thick shaggy deposits of fibrinogen in the basement membrane zone, which is characteristic of LP and occurs in 55% of patients.
2 In our patient, the histopathologic findings of hyperkeratosis, sawtooth configuration of rete ridges, hypergranulosis, vacuolar degeneration, Max Joseph cleft, and lymphohistiocytic inflammatory infiltrate, and the DIF findings of thick shaggy fibrinogen deposits in the basement membrane zone confirmed our clinical and dermatoscopic diagnosis of LP.
Because of its varying clinical presentation, LP can sometimes be confused with other inflammatory disorders like pityriasis rosea, guttate psoriasis and pityriasis lichenoides chronica. Pityriasis rosea is characterized by salmon-colored papules. The lesions are symmetrically distributed and are commonly found on the thoracic, abdominal, and back areas, as well as on the adjoining areas of the neck and extremities.
1 Oral lesions of various types, such as erythematous plaques and ulcers, have been reported to be present in pityriasis rosea.
14 Guttate psoriasis is commonly preceded by a streptococcal throat infection, something that our patient denied experiencing prior to the onset of his lesions. The lesions in guttate psoriasis appear as eruption of small papules with fine white scales, and these lesions are fairly distributed over the trunk and proximal extremities.
15 This condition is also associated with nail changes and pruritus. Pityriasis lichenoides chronica presents as crops of erythematous papules with fine scales that spontaneously regress over weeks to months.
2 Lesions are scattered but discrete, and are usually distributed on the trunk, buttocks, and proximal extremities.
4
Dermatoscopy can be very helpful in narrowing down the differential diagnosis of papulosquamous lesions. Under the dermatoscope, the lesion of pityriasis rosea will have a central mixed vascular pattern and a peripheral collarette of scales.
16 In guttate psoriasis a dermatoscopic pattern of diffuse dotted vessels can be appreciated.
17 Dermatoscopy of pityriasis lichenoides chronica lesions will show irregular linear vessels.
17 The presence of Wickham striae is the distinguishing dermatoscopic feature that differentiates LP from other papulosquamous conditions.
18 19 20
LP has also been linked with liver diseases such as autoimmune chronic active hepatitis, primary biliary cirrhosis and postviral chronic active hepatitis.
2 Hepatitis C infection has been found to be a trigger event in 16 to 29% of patients with LP.
21 Based on epidemiological studies, oral LP has an estimated malignant transformation rate of 0.27% yearly.
22 However, there has been controversy on whether oral LP does indeed lead to malignancy.
23 Existing studies on this are inconsistent in terms of the diagnostic criteria used to diagnose oral LP and the subsequent malignancy, risk factors investigated for malignant transformation, and the management approach to patients with oral LP to ensure early diagnosis of malignancy.
23
Since the severity of symptoms, number and distribution of lesions, and treatment response vary greatly, the treatment of LP is often individualized. Majority of patients with cutaneous LP undergo spontaneous remission after one year.
24 Early or localized lesions of patients with cutaneous LP usually respond to potent topical steroids, such as clobetasol propionate, applied thinly on the lesions twice daily for two to three weeks.
24 Generalized cutaneous LP often needs systemic therapy with oral prednisone 30-60 mg daily for at least four weeks with appropriate tapering.
24 Systemic steroids may help alleviate symptoms, but therapy may not affect the total duration of the disease.
24 Topical medium-potency corticosteroids (e.g., triamcinolone acetonide), high-potency fluorinated corticosteroids (e.g., fluocinolone acetonide, disodium betamethasone phosphate), and—more recently—superpotent halogenated corticosteroids (e.g., clobetasol with adhesive base) are considered first line therapy for oral LP.
25 Oral candidiasis is a complication during therapy, hence chlorhexidine gluconate mouthwash is recommended during the course of treatment.
1 Injection of intralesional triamcinolone acetonide at the proximal nail fold every 4 weeks is recommended for nail lesions.
2
Relapse rate for LP lesions within 3 years after therapy can be as high as 25%.
26 Compared to cutaneous lesions, mucosal membrane lesions may be more resistant to treatment.
4 Nail lesions are difficult to treat, and relapse can be expected even with intralesional corticosteroid administration.
27
In summary, a 56-year-old male with sparse cutaneous papules and macules, and mucosal plaques with erosions of 6 months’ duration came to us. While we considered several papulosquamous dermatoses, our physical examination findings and dermatoscopic findings taken together pointed to atypical LP as the diagnosis, and we were able to start the patient’s therapy on initial consultation. The histologic findings, which came later, was able to confirm our initial diagnosis. Our patient’s condition improved with initial superpotent topical corticosteroids and chlorhexidine gluconate mouthwash, followed by an 8-week course of oral corticosteroids. Dermatoscopy is a non-invasive procedure that is helpful in the assessment of inflammatory dermatoses, such as LP. The use of a clinic dermatoscope and identification of the characteristic Wickham striae of LP led to the early diagnosis and timely management of our patient’s condition, and we did not have to wait for histopathologic confirmation to initiate therapy.
In essence
Lichen planus (LP) is a rare papulosquamous dermatosis that can be diagnosed with the aid of dermatoscopy.
A 57-year-old male patient presented with a 6-month history of sparse, erythematous, cutaneous papules and macules, and mucosal plaques with erosions. We diagnosed the patient as having atypical LP after we saw Wickham striae on some of his lesions using a dermatoscope.
The patient’s cutaneous and mucosal lesions responded to an initial course of topical corticosteroids and chlorhexidine gluconate mouthwash, followed by an 8-week course of oral prednisone.
Acknowledgments
We would like to thank Dr. Ma. Jasmin J. Jamora from the Section of Dermatology in St. Luke’s Medical Center for preparing the direct immunofluorescence report and for providing the images of the said report.
Competing interests
None declared
Access and license
This is an Open Access article licensed under the Creative Commons
Attribution-NonCommercial
4.0 International License, which allows
others to share and adapt the work, provided that derivative works
bear appropriate citation to this original work and are not used for
commercial purposes. To view a copy of this license, visit
http://creativecommons.org/licenses/by-nc/4.0/
References
1. James WD, Berger T, Elston D. Andrew’s Diseases of the Skin: Clinical Dermatology. 12th ed. St. Louis, MO: Saunders Elsevier; 2016.
2. Goldsmith LA, Katz SI, Gilchrest BA, Paller AS, Leffell DJ, Wolff K. Fitzpatrick’s Dermatology in General Medicine. 8th ed. New York: McGraw-Hill; 2012.
3. Zakrzewska JM, Chan ES, Thornhill MH. A systematic review of placebo-controlled randomized clinical trials of treatments used in oral lichen planus. Br J Dermatol. 2005 Aug;153(2):336-341.
4. Usatine RP, Tinitigan M. Diagnosis and Treatment of Lichen Planus. Am Fam Physician. 2011 Jul 1;84(1):53-60.
5. Philippine Dermatological Society Health Information Systems [data file]. Philippine Dermatological Society. 2011 [updated 2017 Feb 9; cited 2017 Feb 17].
6. Friedman P, Sabban EC, Marcucci C, Peralta R, Cabo H. Dermoscopic findings in different clinical variants of lichen planus. Is dermoscopy useful? Dermatol Pract Concept. 2015;5(4):51–55.
7. Clark C. Lichen planus and its management. The Pharmaceutical Journal [Internet]. 2010 Jun 10. Available from: http://www.pharmaceutical-journal.com/learning/learning-article/lichen-planus-and-its-management/11013739.article. Accessed 2 May 2017.
8. Weston G, Payette M. Update on lichen planus and its clinical variants. Inter J of Women’s Dermatology. 2015 Aug;1(3):140-149.
9. Gorouhi F, Davari P, Fazel N. Cutaneous and mucosal lichen planus: a comprehensive review of clinical subtypes, risk factors, diagnosis, and prognosis. The Scientific World Journal. 2014 Jan 30.
10. Tonsager M, Crutchfield CE. Atrophic lichen planus. Dermatol Nurs. 2004;16(1).
11. Haeberle MT, Callen JP, authors. Pityriasis Lichenoides Clinical Presentation [Internet]. Medscape; c1994-2017 [updated 17 Apr 2017]. Available from: http://emedicine.medscape.com/article/1099078-clinical#showall. Accessed 2 May 2017.
12. Lallas A, Argenziano G. Dermatoscope-the dermatologist's stethoscope. Indian J Dermatol Venereol Leprol. 2014 Nov-Dec;80(6):493-494.
13. Rapini RP. Practical Dermatopathology. 2nd ed. Edinburgh: Elsevier; 2012.
14. Vidimos AT, Camisa C. Tongue and cheek: oral lesions in pityriasis rosea. Cutis.
1992 Oct;50(4):276-280.
15. Vence L, Schmitt A, Meadows CE, Gress T. Recognizing Guttate Psoriasis and Initiating Appropriate Treatment. W V Med J. 2015 Jul-Aug;111(4):26-28.
16. Hossam D, Sadek A, Saied N. Dermoscopy: A Literature Review. Egyptian Dermatology Online Journal. 2015 June;11(1):1.
17. Errichetti E, Lacarrubba F, Micali G, Piccirillo A, Stinco G. Differentiation
of pityriasis lichenoides chronica from guttate psoriasis by dermoscopy. Clin Exp
Dermatol. 2015 Oct;40(7):804-806.
18. Tan C, Min ZS, Xue Y, Zhu WY. Spectrum of dermoscopic patterns in lichen planus: a case series from China. J Cutan Med Surg. 2014 Jan-Feb;18(1):28-32.
19. Vásquez-López F, Gómez-Diez S, Sánchez J. Dermoscopy of active lichen planus. Arch Dermatol. 2007;143(8):1092.
20. Zalaudek I, Argenziano G. Dermoscopy subpatterns of inflammatory skin disorders. Arch Dermatol. 2006;142(6):808.
21. Norman RA, Eng W. Clinical Cases in Infections and Infestations of the Skin. 2015 ed. Switzerland:Springer; 2015.
22. Garcia de Souza F, Paradella T. Malignant potential of oral lichen planus: a
meta-analysis. Rev Odonto Ciência. 2009;24:194–197.
23. Gonzalez-Moles MA, Scully C, Gil-Montoya JA. Oral lichen planus: controversies surrounding malignant transformation. Oral Diseases. 2008;14(3):229-243.
24. Rajani K. Lichen Planus. Am Fam Physician. 2000 Jun 1;61(11):3319-3324.
25. Lavanya N, Jayanthi P, Rao UK, Ranganathan K. Oral lichen planus: an update on pathogenesis and treatment. J Oral Maxillofac Pathol. 2011 May-Aug;15(2):127–132.
26. Cribier B, Frances C, Chosidow O. Treatment of lichen planus an evidence-based medicine analysis of efficacy. Arch Dermatol. 1998;134(12):1521-1530.
27. Lehman JS, Tollefson MM, Gibson LE. Lichen planus. Int J Dermatol. 2009 Jun;48(7):493-494.
Copyright © 2017 JKM Martinez, et al.






